Brief intro from Dr. Rollins
Ten years ago we sat in a motel room in Sweetwater with a team of 10 scientists from Texas A&M, Texas A&M – Kingsville, Texas Tech University, and the University of North Texas. We had been given a charge from Rick Snipes, President of the Rolling Plains Quail Research Foundation. Our charge was to design and conduct a comprehensive scientific investigation into disease (in the broadest context) as a possible explanation for the decline of bobwhites in the Rolling Plains. Over the next 24 hours, we pondered pathogens and parasites, and the procedures and collaborations that would be necessary to pull it off.
RPQRF’s Position Statement on Eyeworms & Cecal Worms
(revised 1-23)
Introduction
- Researchers at RPQRR have investigated infections of helminths (parasitic worms) in bobwhites since 2009. Results from our “Operation Idiopathic Decline” initiative subsequently prompted much interest, effort, and dollars in subsequent studies.
- Research on eyeworms and cecal worms, and their potential impacts on quail populations, can be contentious. The following position statement has been approved by RPQRF’s Board of Directors.
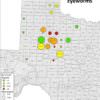
- Historical background- Operation Idiopathic Decline canvassed 35 counties across west Texas and western Oklahoma, see Figure 1. Bobwhite and scaled quail were trapped in Aug and Oct 2011-13 and birds sampled for various pathogens. The prevalence of eyeworms and cecal worms, see Figure 2, across these sites prompted subsequent research to determine possible impacts on quail populations.
- Key terms used in this discussion
- Prevalence – proportion of individuals in a population that are infected by a species of parasite; expressed as percent.
- Intensity – the number of parasites of a given species (e.g., eyeworms) for only the infected subset of individuals in a population.
- Example: If ten bobwhites are examined for eyeworms and half are infected (n =5), then prevalence is 50% (5/10). If each infected bobwhite has 7 worms, the average intensity is 7 worms per bird.
What We Know: Eyeworms
- Eyeworms were first documented infecting bobwhites in 1959 in Cottle County (Matador wildlife Management Area).
- Infection rates reported were similar (e.g., prevalence, intensity) as seen in contemporary studies. (source: reports from TPWD from the Matador WMA));
- Subsequent research on helminths (parasitic worms) in quails mostly failed to investigate the importance of eyeworms as researchers tended to examine only the intestines and ignore the eyes (especially the tissues behind the eye). Hence, these early reports were conservative estimates of prevalence and intensity.
- Eyeworms are 8X more prevalent in bobwhite from Rolling Plains vs South Texas Plains;
- Since 2015 the RPQRR has monitored eyeworm dynamics in bobwhites across multiple counties in Texas.
- Prevalence, abundance, and intensity of eyeworms in the Rolling Plains varies year-to-year and ranch-to-ranch (Figure, table). Average prevalence during 2017-2022 from all Texas submissions received was 61% with 13 worms per bird.
- Bobwhite accumulate eyeworms over time. Thus, prevalence and intensity are higher in adults than juveniles. “Poor” quail seasons (e.g., 2019) will yield higher eyeworm prevalence and intensities because few juvenile quail are present in the population.
- Eyeworm presence may cause keratitis (scarring of the cornea) and other symptoms indicative of inflammation.
- As many as 24 potential intermediate hosts have been identified including cockroaches, field crickets, and several species of grasshoppers which are all key food components for quail.
- New molecular techniques are available to allow for on-site, non-lethal sampling of quail to detect eyeworm infection.
- Infections in scaled quail are typically about 25% of those observed in bobwhites.
What We Know: Cecal Worms
- Across their range, bobwhites are parasitized by numerous species of cecal nematodes, or roundworms, in the middle part of the quail’s gut (ceca). The most frequently occurring cecal worm in the Rolling Plains and South Texas is A. pennula.
- Prevalence and intensity vary by year and site. On average, prevalence of A. pennula is approximately 85% for bobwhite in the Rolling Plains, typically less in south Texas.
- A. pennula is an “imbibing” (i.e., free-floating) nematode that has not been documented to cause necrosis or penetration of the cecal wall or direct immune response in bobwhite.
- Thirteen different species of grasshoppers have been identified as potential intermediate hosts.
- New molecular techniques are available to allow for on-site, non-lethal sampling of quail to detect cecal worm infections.
- See: https://www.quailresearch.org/resources/#faqq for more on “what are cecal worms?”
What We Don’t Know (at this point)
- While bobwhite and scaled quail in the Rolling Plains are infected at a seemingly high prevalence and intensity, it has not been documented at this time whether parasites are a limiting factor for quail populations.
- There is currently much speculation as to the effect of helminths (parasitic worms) on bobwhite and scaled quail populations. At this time, these should be considered hypotheses neither supported nor refuted by independent, replicated scientific studies.
- We do not know the threshold at which infections begin to manifest negative consequences (if any) on survival and reproduction, by whatever ecological mechanism.
- Additional research is needed to understand the impact, if any, that these worms have at the population level in order to assess the impact(s) of these nematodes as potential factors in quail decline.
Where We’re Headed
- RPQRF will remain objective studying the effects of helminths of quail
populations. Future research will focus on effects of parasites on demographics (e.g., survival
and reproduction).
Related Webisodes
- Monitoring Quail for Eyeworms
(https://www.youtube.com/watch?v=QhcioPX8pOw&t=505s) - Addressing the Eyeworm Threat in Quail
(https://www.youtube.com/watch?v=tSN0Zi75Cy0&t=95s) - Cecal Worms in Quail
(https://www.youtube.com/watch?v=fjfKW5LrhBU&t=86s) - Operation Idiopathic Decline: Search for the Smoking Gun
(https://www.youtube.com/watch?v=Djo5gzDjA2w)
Publications by Other Authors Supported by RPQRF:
Bedford, K. 2015. Parasitological survey of scaled quail from west Texas. Thesis. Texas A&M University, Kingsville.
Bruno, A., A. M. Fedynich, A. Smith-Herron, and D. Rollins. 2015. Pathological response of northern bobwhites to Oxyspirura petrowi infections. Journal of Parasitology 101:364–368.
Bruno, A., A.M. Fedynich, D. Rollins, and D.B. Wester. 2018. Helminth community and host dynamics in northern bobwhites from the Rolling Plains Ecoregion, U.S.A. Journal of Helminthology 1–7. https://doi.org/10.1017/S0022149X18000494
Ferro, P.J., O. Khan , C. Vuong , S. M. Reddy , L. LaCoste , D. Rollins , and B. Lupiani. 2012. Avian Influenza Virus Investigation in Wild Bobwhite Quail from Texas. Avian Diseases, 56:858-860.
Kubečka, B.W., A. Bruno, and D. Rollins. 2017. Geographic Survey of Oxyspirura petrowi Among Wild Northern Bobwhites in the United States,” National Quail Symposium Proceedings: Vol. 8, Article 84. Available at: http://trace.tennessee.edu/nqsp/vol8/iss1/84
Kubecka, B.W., N. J. Traub, V. V. Tkach, T.R. Shirley, D. Rollins, and A. M. Fedynich. 2018. Mesocestoides sp. in Wild Northern Bobwhite (Colinus virginianus) and Scaled Quail (Callipepla squamata). Journal of Wildlife Diseases, 54(3): In press.
Su, H., J. McKelvey, D. Rollins, M. Zhang,, D.J. Brightsmith, et al. 2014. Cultivable Bacterial Microbiota of Northern Bobwhite (Colinus virginianus): A New Reservoir of Antimicrobial Resistance? PLoS ONE 9(6): e99826. doi:10.1371/journal.pone.0099826
Villarreal, S.M., Fedynich, A., L. A. Brennan, and D. Rollins. (2012) “Parasitic Eyeworm (Oxyspirura petrowi) in Northern Bobwhites from the Rolling Plains of Texas, 2007–2011,” National Quail Symposium Proceedings: Vol. 7, Article 95.
Villarreal, S.M., A. Bruno, A.M. Fedynich, L.A. Brennan, and D. Rollins. 2016. Helminth Infections Across a Northern Bobwhite (Colinus virginianus) Annual Cycle in Fisher County, Texas. Western North American Naturalist 76(3):275-280.
Parasite FAQ
How do quail become infected with eyeworms?
Quail must consume an infected “intermediate host” (e.g., grasshopper, cricket, cockroach). Once inside the quail’s crop, eyeworm larvae from the intermediate host escape, make their way into the esophagus, and move up to the lacrimal ducts, then migrate up into the eye. This process can occur in less than 30 minutes.
Are these eyeworms only in quail?
No, they have been found in pheasants, lesser prairie chickens, mockingbirds, and several other species. Interestingly, we surveyed for their presence in wild turkeys on a site that had “high” infection rates of eyeworms. While the turkeys were eating arthropods (presumably infected with eyeworm larvae), only 1 of 104 turkeys had an eyeworm.
Is the eyeworm native to North America?
Parasites aren’t typically classified as native or exotic. Skrjabin (1929) first characterized O. petrowi in Northeastern Europe, and early reports in the U.S. date back to the 1940s.
I've only heard of this in Texas—is it limited to there or does it occur elsewhere?
While the Rolling Plains ecoregion of Texas appears to have the highest infection rates, we have also found them in bobwhites from AL, OK, and VA. They likely occur in KS and NE but we have not examined birds from those states. See Geographic Survey of Oxyspirura Petrowi Among Wild Northern Bobwhites in the United States.
Do eyeworms cause blindness in an infected quail?
They can irritate (i.e., cause inflammation) to the optic nerve and associated glands situated behind the eyes. They can also produce scarring of the cornea. Such pathology likely affects the quail’s vision. Odds are it would be preyed upon before it became “blind.”
What percentage of bobwhites have eyeworms?
Since we began studying them in 2009, we typically find that 50-60 percent of the bobwhites harbor eyeworms. Some sites have upwards of 80 percent infection. Infections in the Rolling Plains are about 8X greater than bobwhites from south Texas. Adult quail typically have greater infections than juvenile birds.
Are the birds okay for human consumption if they have eyeworms?
Yes, no concerns for human consumption . . . most of us don’t eat the heads anyway!
How do I know if my quail have eyeworms?
For initial inspection in the field, see Joe Crafton’s technique above (https://youtu.be/VSfwWONuU0Y ). Also watch the video “How to Search for Eyeworms in Quail”. Be aware however that both of these techniques underestimate the number of eyeworms, as many (probably most) reside behind the eye itself. A complete dissection of the head is needed to get an accurate count.
So do you think eyeworms are worse now than in times past or is it just that we are aware that they exist now and know how to check for them?
We’ve known about eyeworms since early 1960s in bobwhites; they were noted in pheasants in the Midwest earlier than that. Information about them in quail essentially lay “dormant” for the past 50 years, because most parasite studies don’t look at/in/around the eyes–looking typically in the GI tract. Our “Operation Idiopathic Decline” study (initiated in 2011; see video at https://www.youtube.com/watch?v=xUfqw8dGk5E ) resulted in newfound interest in eyeworms.